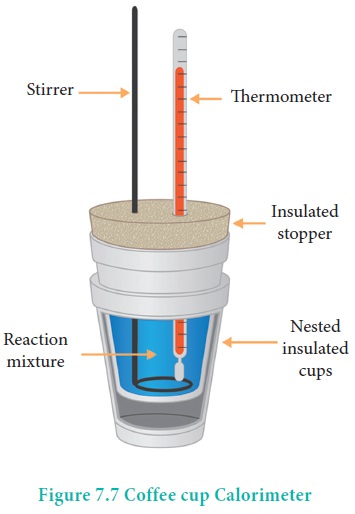
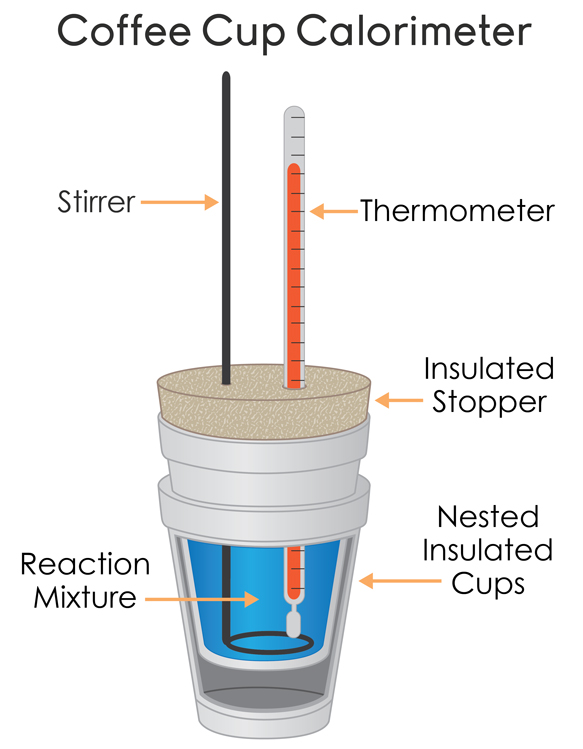
In most everyday situations, the change in enthalpy occurs at constant pressure, so any enthalpy changes are linked to the heat absorbed or produced by the reaction.Study with quizlet and memorize flashcards containing terms like when 40.0 ml of 1.00 m h2so4 is added to 80.0 ml of 1.00 m naoh at 20.00°c in a coffee cup calorimeter, the temperature of the aqueous solution increases to 29.20°c.
This section delves into the science behind enthalpy and how heat transfer can be quantified.The calorimetry calculator can help you solve complex calorimetry problems.When 50.0 ml of 0.10 m hcl(aq) and 50.0 ml of 1.00 m naoh(aq), both at 22.0 °c, are added to a coffee cup calorimeter, the temperature of the mixture reaches a maximum of 28.9 °c.
Read on to learn what calorimetry is and how to solve calorimetry problems with the proper equations.The main difference with bomb calorimetry is that specific heat and the weight of the material in the.
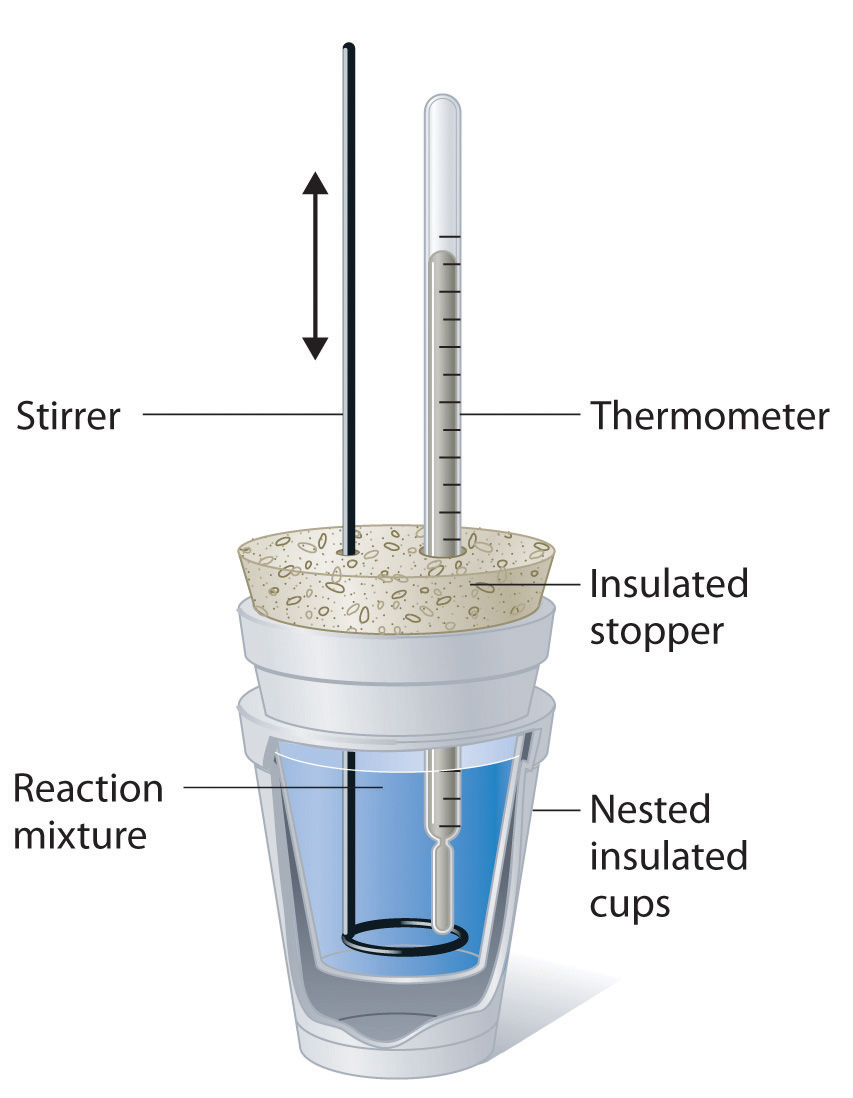
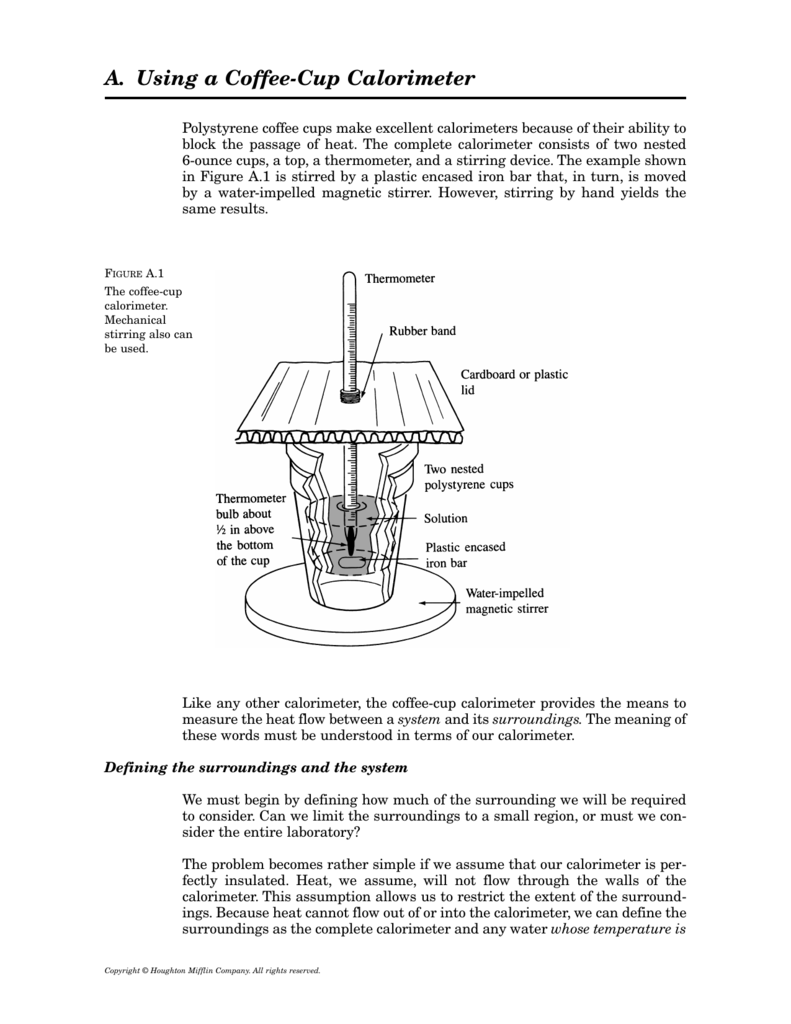
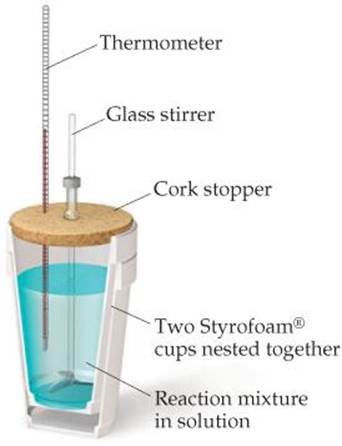
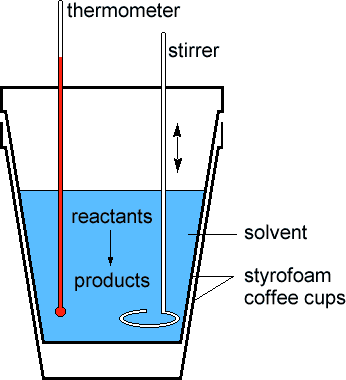
In this technique, a process is carried out in solution in a coffee cup calorimeter, an inexpensive device composed of two styrofoam cups.This text is adapted from openstax, chemistry 2e, section 5.2:If the mass of the solution is 120.0 g and the specific heat of the calorimeter and solution is 4.184 j/g • °c, how much heat is given off in the reaction.
It can analyze the heat exchange between up to 3 objects.The calorimeter is filled with 200.0 grams of water at 22.9 ° c.
The change in enthalpy (δh) is negative because the energy is being lost by the system (the heater) and gained by the surroundings.
Last update images today Coffee Cup Calorimeter
Tom Banton, Tom Abell Battle For Somerset But Warwickshire Turn The Screw
Northamptonshire 97 and 137 for 4 (Gay 67) need a further 147 to beat Sussex 143 (White 4-23, Procter 3-37) and 237 (Simpson 40, Procter 4-45)
Sussex's bowlers dealt Northamptonshire a double blow late in the day at Wantage Road to close in on victory and the chance to stretch their advantage at the top of Vitality County Championship Division Two.
Set a target of 284, Emilio Gay's 67 appeared to have given Northamptonshire a fighting chance - but he and skipper Luke Procter both fell in the space of four balls to leave the visitors as clear favourites in a low-scoring contest.
Seamer Nathan McAndrew, who dismissed Gay leg before, ended with 2 for 37 as the home side closed on 137 for 4 - still needing another 147 to pull off a first win of the season.
Procter played a key role with the ball earlier in the day, recording season's best figures of 4 for 45 as Sussex were bowled out for 237 in their second innings, while Ben Sanderson took 3 for 44.
The opening day's play had encompassed 22 wickets - and more of the same looked on the cards when Tom Alsop departed in the first over, edging Sanderson behind with no increase to Sussex's overnight lead of 129. In a tight opening spell Sanderson soon dismissed nightwatcher Sean Hunt as well, taken at first slip, while Raphy Weatherall and Justin Broad also extracted movement and bounce off the surface.
James Coles settled down to dispatch a series of classic cover drives to the boundary, while Oli Carter shook off a slow start by whipping Procter for six over square leg, but the Northamptonshire captain responded by bringing one back to pin Coles in front.
Broad then struck twice just after lunch with a ball that seamed and bounced to clip Carter's off bail before Fynn Hudson-Prentice miscalculated a cut shot and speared it into the hands of mid-off instead.
Having spilled a number of catches in Sussex's first innings, the home side squandered an opportunity to remove John Simpson as Gay, springing across from second slip, fumbled a sharp chance off Sanderson. In the same over, Broad's throw from cover should have left McAndrew short of his ground and the Australian capitalised on that let-off, thrashing five boundaries to reach a valuable 22 before he was out in peculiar fashion.
McAndrew's leading edge off Procter looped up towards mid-off, with the bowler racing to gather it, colliding with the substitute fielder and dumping the ball on the turf in his follow-through - yet the catch stood.
Procter swiftly polished off the last two wickets, including that of Simpson for 40, to leave Northamptonshire almost a session and a half's batting and they lost Ricardo Vasconcelos prior to tea, trapped in his crease by McAndrew's second delivery.
However, Prithvi Shaw got off the mark by crunching McAndrew to the cover fence and continued to exhibit classy strokeplay on either side of the wicket as he and Gay built a partnership of 69, the highest of the match.
Left-armer Hunt produced a beauty to separate the pair, knocking back Shaw's off stump for 37 - and he might also have dislodged Gay in his next over but Simpson, leaping low to his left, was unable to pouch the chance.
The opener took advantage to pass 50 for the sixth time this season from the next ball and he and Procter dug in to reach 118 for 2 before Sussex prised them both out in successive overs. Offspinner Jack Carson persuaded Procter to nudge one to Simpson and the swift loss of Gay left Matthew Breetzke and Rob Keogh with the task of ensuring Northamptonshire at least live to fight another day.








:max_bytes(150000):strip_icc()/GettyImages-141482586-566245ac3df78ce16197a1fd.jpg)