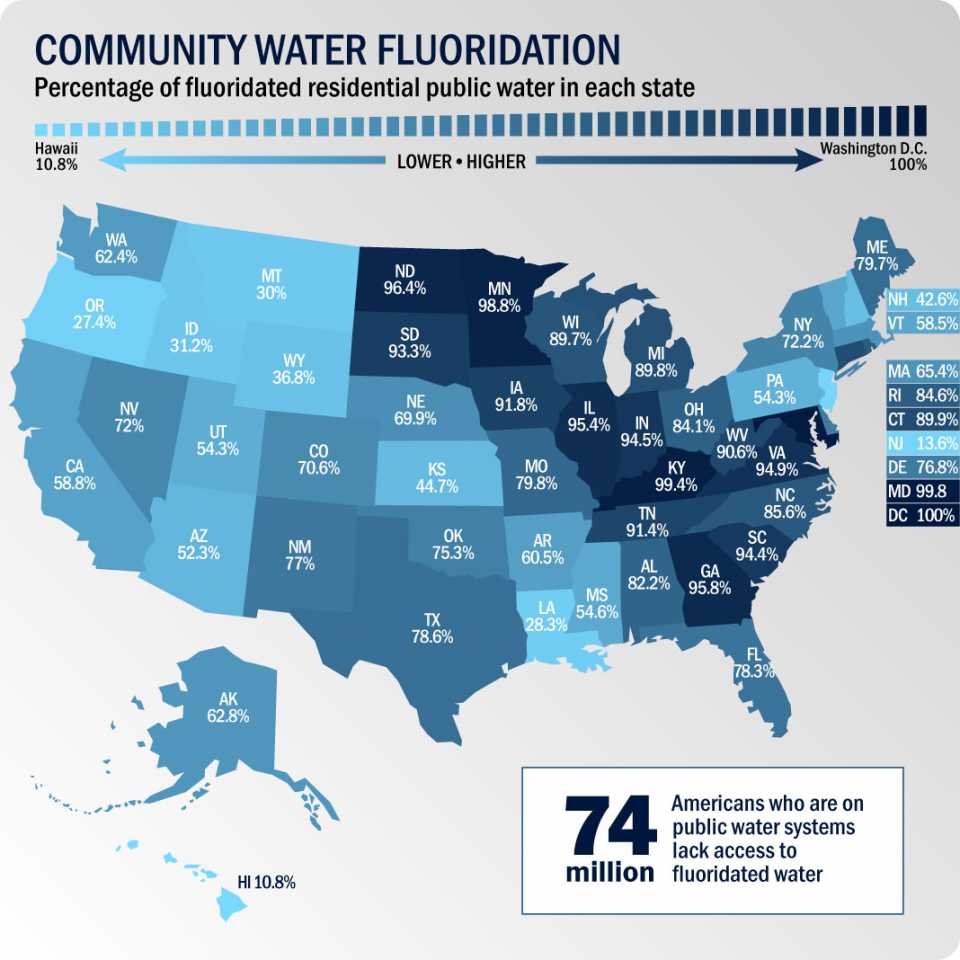
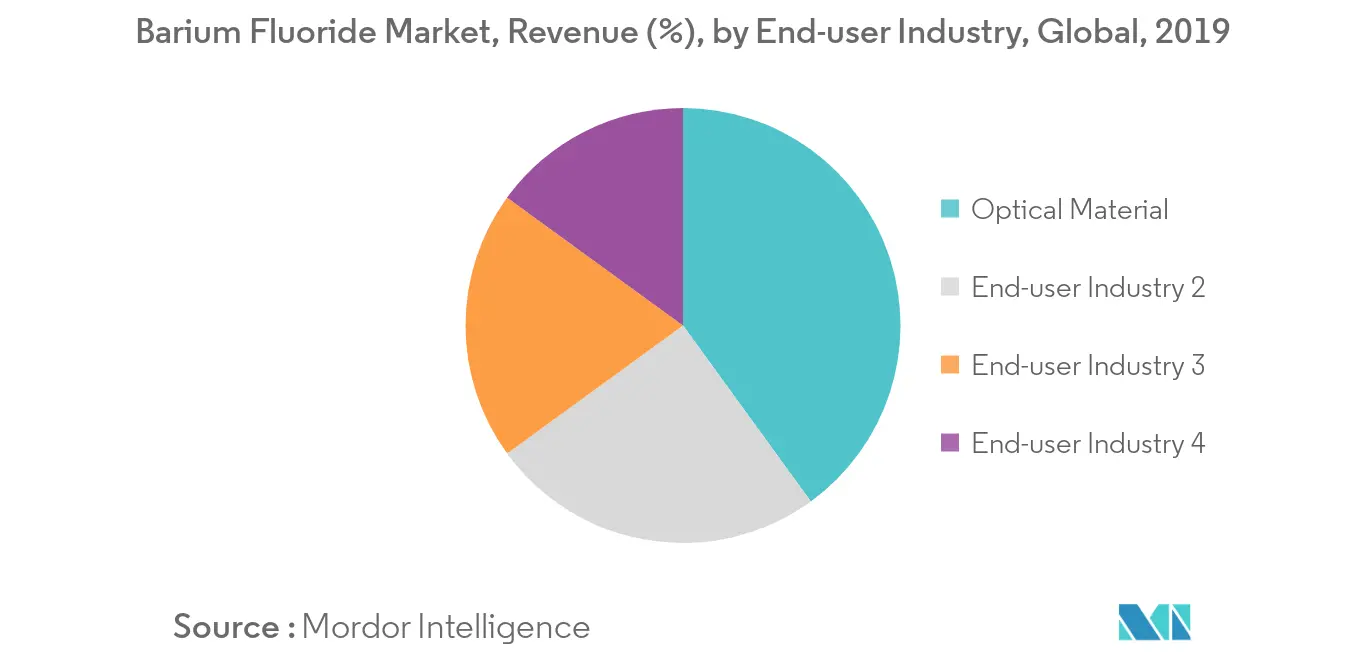
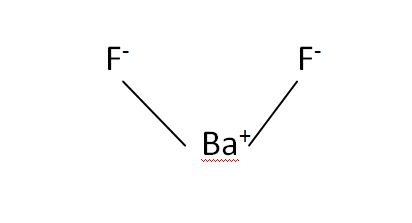
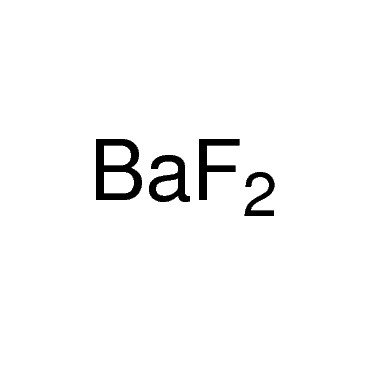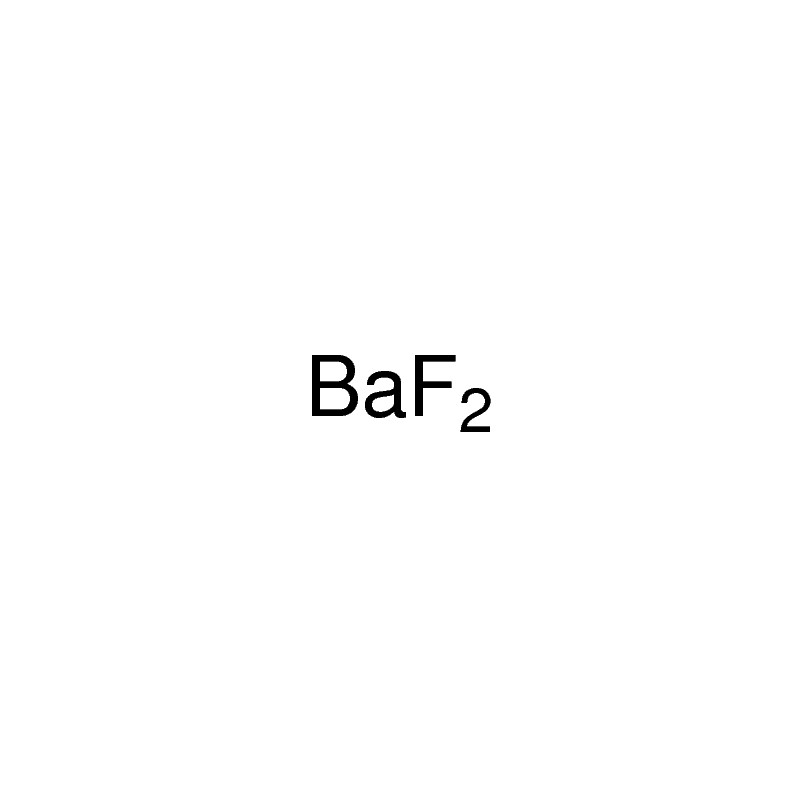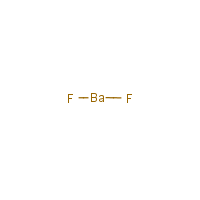Barium, abbreviated as 'ba,' is a group 2 element with atomic number 56 in the periodic chart.Barium fluoride, precipitated, 99.999% trace metals basis.
1 nmr, and 2 raman.Barium fluoride view entire compound with spectra:Like caf 2, it is resilient to and insoluble in water.
Finally, add together the total mass of each element to get the molar mass of ba (f2):Solid (mineral oil mull) instrument:
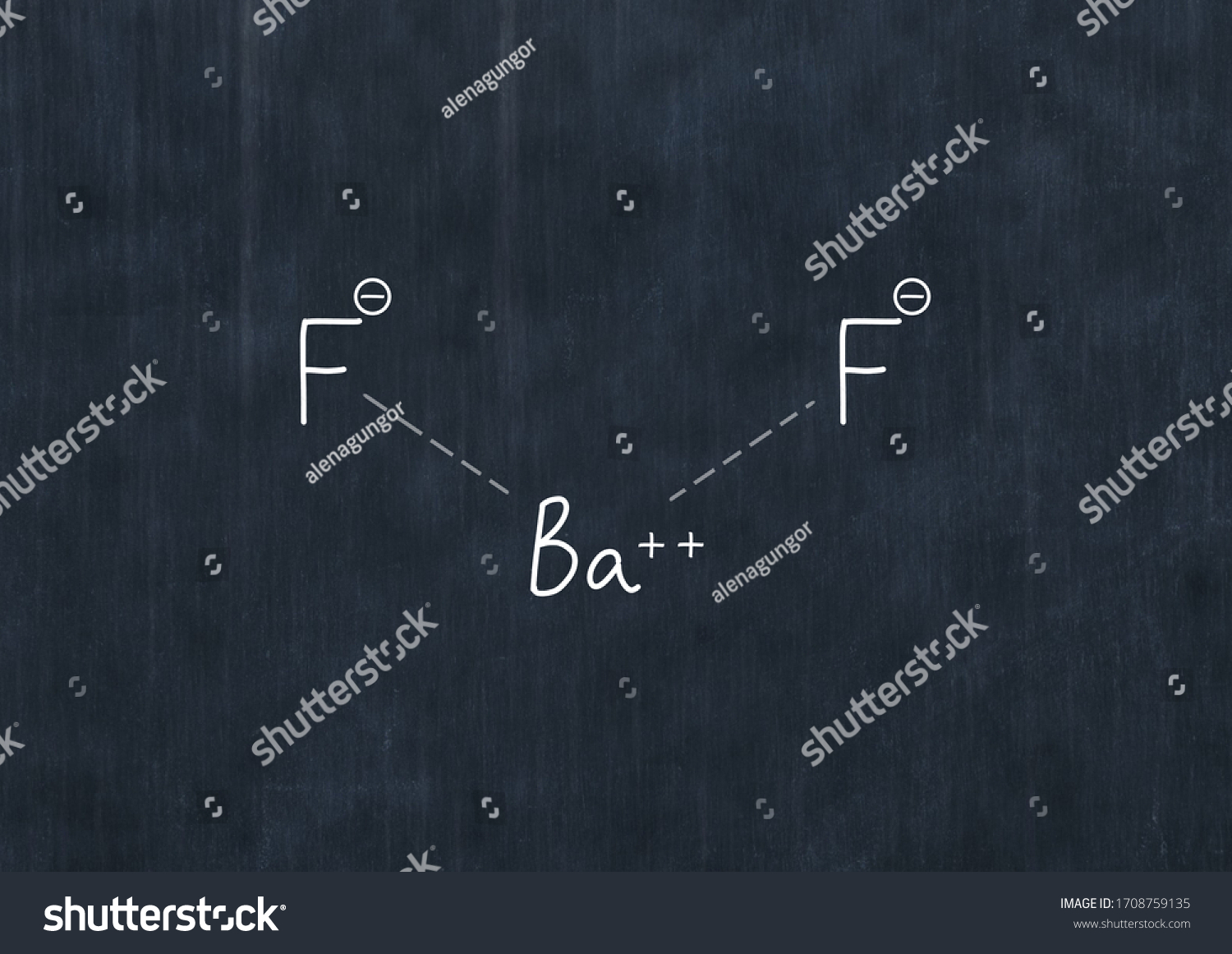
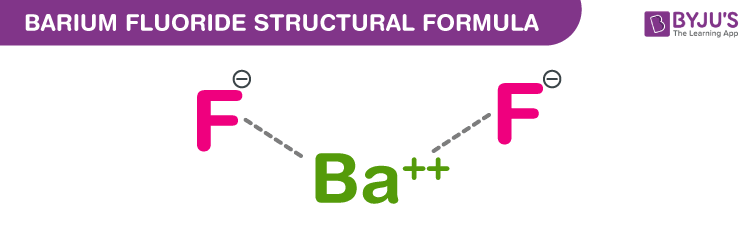
Nist chemistry webbook the national institute of standards and technology (nist) uses its best efforts to deliver a high quality copy of the database and to verify that the data contained therein have been selected on the basis of sound scientific judgment.To find the formula of an ionic compound, first identify the cation and write down its symbol and charge.Thus, this can't be the formula for barium fluoride as one to one ratio of the charges makes the ionic formula.
Barium is a dense alkaline earth metal found in ore deposits and accounts for 0.05 percent of the earth's crust.Usually metals and nonmetals form ionic compounds when chemically combined.
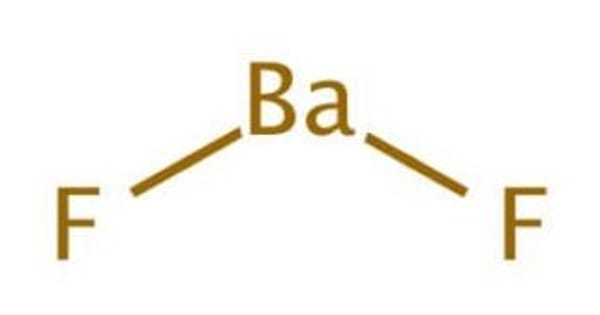
The chemical formula for barium fluoride is baf2.Barium fluoride crystal optic disc, 32mm x 3mm, polished both sides.So hg 2 cl 2 it the lowest whole number ratio of cation to anion.
Under standard conditions it adopts the fluorite structure and at high pressure the pbcl 2 structure.137.327 g/mol + 37.9968064 g/mol = 175.3238064 g/mol.
The full spectrum can only be viewed using a free account.
Last update images today Barium Fluoride Formula Name
 What The Numbers Tell Us About Shakur Stevenson Vs. Artem Harutyunyan
WASHINGTON -- — Jake Irvin allowed just one hit and issued one walk in eight innings, Jesse Winker hit a pinch-hit home run and the Washington Nationals beat the New York Mets 1-0 on Thursday in the traditional 11 a.m. Fourth of July game in the nation’s capital.
Irvin (7-6) pumped his chest and gestured to the crowd for applause after striking out Tyrone Taylor on his 99th pitch and walking off the mound following the longest outing of his major league career. That was Irvin's eighth strikeout, and he faced just one batter over the minimum, retiring the final 17 batters he faced.
“It was a fun day, and (catcher Riley Adams) and I were on the same page all day,” Irvin said. "Being able to pitch on this holiday, it’s something truly special to me, really near and dear to my heart and I loved it, had a lot of fun.”
The Mets were blanked for the seventh time this season and were held to only one hit in a game for the first time since a loss to Milwaukee on opening day, something manager Carlos Mendoza chalked up to Irvin's masterful performance.
"We had a hard time catching up to his fastball," Mendoza said. “I thought we were late at times for his fastball, and then the way he used the breaking ball kept us off balance, so we couldn’t get much going.”
Winker made sure Irvin left with the victory by hitting a curveball from Adrian Houser 407 feet to right-center field. It was his fifth career pinch-hit home run and his 10th of any kind this season.
“He was biting at the bit, I can tell you that," Nationals manager Dave Martinez said. "He said, ‘I’ll be ready.’ I said, ‘Oh, I know you’re going to be ready.’ But to come up here and do what he did, it’s awesome. Big win. Big win for us.”
Houser (1-5) became the latest Mets reliever to give up a big hit as the beleaguered bullpen slogs on without suspended closer Edwin Díaz.
Before Winker's home run, the game was a pitcher's duel — one New York wasted for left-handed starter Jose Quintana, who allowed four hits, walked three and struck out one over seven scoreless innings. He gave the Mets the kind of start they needed given the amount of work the relievers have had to put in with Díaz unavailable.
“It means a lot, especially the state of the bullpen," Quinta said. “If you can save one, two innings (for) the bullpen, that’s really good for us.”
Derek Law tossed a perfect ninth inning for his first save of the season and the Nationals' second consecutive win, while the Mets fell back below .500 at 42-43.
“It happens," Mendoza said. "Just got to turn the page and be ready for another series.”
TRAINER'S ROOM
Mets: RHP Kodai Senga (shoulder/triceps) is scheduled to throw roughly 50 pitches in his next minor league rehab start next week, which Mendoza said will likely be Tuesday with Triple-A Syracuse.
UP NEXT
Mets: Unless rain derails it, the series opener Friday at Pittsburgh features RHP Luis Severino (5-2, 3.42 ERA) against Pirates rookie RHP Paul Skenes (4-0, 2.06).
Nationals: Continue their homestand Friday against St. Louis, with LHP Patrick Corbin (1-8, 5.49) starting against Cardinals RHP Sonny Gray (9-5, 2.98).
------
AP MLB: https://apnews.com/hub/mlb
What The Numbers Tell Us About Shakur Stevenson Vs. Artem Harutyunyan
WASHINGTON -- — Jake Irvin allowed just one hit and issued one walk in eight innings, Jesse Winker hit a pinch-hit home run and the Washington Nationals beat the New York Mets 1-0 on Thursday in the traditional 11 a.m. Fourth of July game in the nation’s capital.
Irvin (7-6) pumped his chest and gestured to the crowd for applause after striking out Tyrone Taylor on his 99th pitch and walking off the mound following the longest outing of his major league career. That was Irvin's eighth strikeout, and he faced just one batter over the minimum, retiring the final 17 batters he faced.
“It was a fun day, and (catcher Riley Adams) and I were on the same page all day,” Irvin said. "Being able to pitch on this holiday, it’s something truly special to me, really near and dear to my heart and I loved it, had a lot of fun.”
The Mets were blanked for the seventh time this season and were held to only one hit in a game for the first time since a loss to Milwaukee on opening day, something manager Carlos Mendoza chalked up to Irvin's masterful performance.
"We had a hard time catching up to his fastball," Mendoza said. “I thought we were late at times for his fastball, and then the way he used the breaking ball kept us off balance, so we couldn’t get much going.”
Winker made sure Irvin left with the victory by hitting a curveball from Adrian Houser 407 feet to right-center field. It was his fifth career pinch-hit home run and his 10th of any kind this season.
“He was biting at the bit, I can tell you that," Nationals manager Dave Martinez said. "He said, ‘I’ll be ready.’ I said, ‘Oh, I know you’re going to be ready.’ But to come up here and do what he did, it’s awesome. Big win. Big win for us.”
Houser (1-5) became the latest Mets reliever to give up a big hit as the beleaguered bullpen slogs on without suspended closer Edwin Díaz.
Before Winker's home run, the game was a pitcher's duel — one New York wasted for left-handed starter Jose Quintana, who allowed four hits, walked three and struck out one over seven scoreless innings. He gave the Mets the kind of start they needed given the amount of work the relievers have had to put in with Díaz unavailable.
“It means a lot, especially the state of the bullpen," Quinta said. “If you can save one, two innings (for) the bullpen, that’s really good for us.”
Derek Law tossed a perfect ninth inning for his first save of the season and the Nationals' second consecutive win, while the Mets fell back below .500 at 42-43.
“It happens," Mendoza said. "Just got to turn the page and be ready for another series.”
TRAINER'S ROOM
Mets: RHP Kodai Senga (shoulder/triceps) is scheduled to throw roughly 50 pitches in his next minor league rehab start next week, which Mendoza said will likely be Tuesday with Triple-A Syracuse.
UP NEXT
Mets: Unless rain derails it, the series opener Friday at Pittsburgh features RHP Luis Severino (5-2, 3.42 ERA) against Pirates rookie RHP Paul Skenes (4-0, 2.06).
Nationals: Continue their homestand Friday against St. Louis, with LHP Patrick Corbin (1-8, 5.49) starting against Cardinals RHP Sonny Gray (9-5, 2.98).
------
AP MLB: https://apnews.com/hub/mlb